Endomembrane System
What is the Endomembrane System
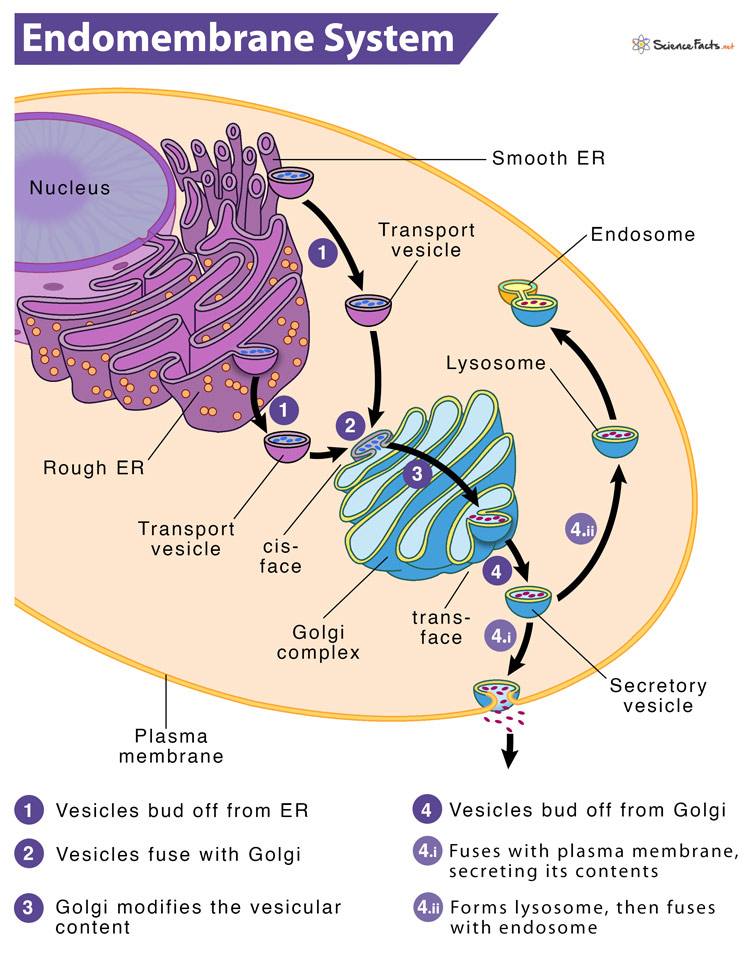
The endomembrane (endo means within) system is a set of membranes in eukaryotic cells spreading from the nuclear membrane to the plasma membrane. These membranes divide the cell into different functional and structural compartments or organelles. The components of the system work together to modify, package and transport lipids and proteins.
The system is defined as a set of membranes that give rise to a single functional and developmental unit, either directly connected or exchanging material through vesicular transport.
Function: What it Does in a Cell
The endomembrane system is an internal membrane system within the cell that carries out a variety of functions such as:
- Synthesis of proteins and their transport
- Metabolism and movement of lipids
- The detoxification of poison
Here, the membranes are either directly in contact with each other or can communicate through the formation of vesicles.
Components: Which Organelles are Part of the Endomembrane System
The endomembrane system comprises seven different parts or components of a typical eukaryotic cell. They are sequentially listed below in the order of protein and lipid secretion in a cell.
- The Nuclear Envelope
- The Endoplasmic Reticulum
- The Golgi Apparatus
- Lysosomes
- Endosomes
- Transport Vesicles
- The Plasma Membrane
In the subsequent section below, we will deal with each of them and explain how they help regulate proteins and lipids.
1) The Nuclear Envelope
The nuclear envelope is a membranous bilayer covering of the nucleus that separates the contents of the nucleus from the cytoplasm. Each of its membranes is a lipid bilayer with associated proteins. The outer or external nuclear membrane is continuous with the rough endoplasmic reticulum (RER) membrane and has ribosomes attached to the surface.
The two layers fuse at nuclear pores, perforations on the nuclear envelope. These pores measure about 120 nm in diameter and regulate the passage of molecules in and out of the nucleus. The space between the external and internal membranes is known as perinuclear space, which is continuous with the lumen of RER. The nuclear membrane of a typical mammalian cell contains about 3000–4000 pore complexes. These pores are highly efficient at selectively allowing materials in and out of the nucleus since the nuclear envelope has a considerable amount of traffic.
Protein synthesis begins in the nucleus. At first, the DNA segment that encodes a particular protein gets copied by a process called transcription, producing a ‘transcript’ in the form of mRNA.
Then, the newly synthesized mRNA exits the nucleus through the nuclear pore guided by the export receptors in the nuclear membrane. Upon reaching the cytosol, the nuclear export signal is removed from the mRNA, and then it returns to the nucleus.
In the cytosol, the ribosome identifies the mRNA and translates the code into amino acids with the help of matching tRNAs.
After translation, the newly-formed protein is transported to the endoplasmic reticulum.
2) The Endoplasmic Reticulum (ER)
ER consists of an interconnected network of flattened sacs and branching tubules. It divides the cytoplasm into two distinct compartments: the hollow space inside the discs and tubules called the lumen or cisternal space and the extraluminal space. The ER plays a vital role in the synthesis of lipids and the modification of proteins.
Depending on the attachment of ribosomes, ER is of two types. They are as follows:
Rough Endoplasmic Reticulum (RER)
The rough endoplasmic reticulum (RER) gets its name from the bumpy ribosomes attached to its cytoplasmic surface, giving it a studded appearance.
Ribosomes synthesize proteins and transfer them into the lumen of the RER, where they go through post-translational modifications, such as folding or adding carbohydrate side chains. This phenomenon is also called ‘post-translational translocation.’ Another critical event called ‘cotranslational translocation’ occurs when the ribosome takes the initial chain of 16-30 amino acids (peptide) recognized by the signal recognition particle or signal peptide to the ER even without finishing the translation yet.
The ribosome, along with its peptide cargo, moves to the ER and binds to its surface via the binding site called translocon, with the help of GTP molecules that strengthens the interaction. This binding activates ER membrane protein complex to form a passageway or translocation channel through which the newly synthesized peptide can travel and reach the ER lumen. The ribosome, then, resumes translating the mRNA.
With the further addition of amino acids to the signal peptide, the peptide is pushed into the ER lumen through the translocation channel. After translation, the whole protein eventually gets released into the ER lumen. The signal peptide gets cleaved off by a signal peptidase, and the nascent protein then undergoes maturation.
These modified proteins are either incorporated into cellular membranes, like ER membrane, or secreted from the cell in the form of hormones or enzymes.
Suppose the modified proteins are not meant to stay in the ER. In that case, they are packaged into tiny spheres of membrane called vesicles and shipped to the Golgi for further processing and modification.
The RER also synthesizes phospholipids for other cellular membranes, which reach their destinations via transport vesicles that bud from the RER’s membrane.
Smooth Endoplasmic Reticulum (SER)
The SER is continuous with the RER but bears few or no ribosomes on its cytoplasmic surface.
It is the chief site of lipid synthesis. The majority of lipids, such as phospholipids, sterols, steroids, ceramides, and triglycerides, get synthesized in the lumen of SER mediated by various enzymes present there.
The SER in muscle cells, called the sarcoplasmic reticulum, regulates intracellular calcium concentration needed to trigger muscle fibers’ coordinated contractions.
SER is also associated with carbohydrate metabolism. It contains the enzyme glucose-6-phosphatase, which converts glucose 6-phosphate into glucose.
It also helps in drug detoxification. For instance, the SER of liver cells contains cytochrome P450s residing in its lumen that helps to detoxify drugs and poisons by adding a hydroxyl group to the drug molecule.
There are also tiny “smooth” patches of ER found within the rough ER. These patches, called transitional ER, serve as exit sites for vesicles budding off from the rough ER.
3) The Golgi Apparatus
Like ER, the Golgi apparatus (also called Golgi complex or Golgi body) is an organelle composed of cisternae (flattened membrane sacs containing fluid) forming a stack. In animal cells, the cisternae are connected by microtubules, whereas, in plant cells, they connect them. Also, unlike the ER, the Golgi cisternae are not directly connected to the nuclear envelope. Nevertheless, the cisternae are derived from the vesicles that bud off from the ER. So, a part of the Golgi apparatus is commonly seen near the exit sites of ER.
Based on the location of the cisternae in the stack, they may be cis, medial, or trans. Each cisterna has specific enzymes anchored in its membrane and therefore involved in specific biological activities. Overall, the cis face contains enzymes for early modifications of proteins. In contrast, the trans face contains enzymes that are involved in the final modifications of protein.
As these cisternae have no fixed positions, they move outward. Thus, the cisternae found closest to the ER is cis face cisternae, the central cisternae are called the medial, and the farthest from the ER is trans face cisternae. It means a cisterna starts as cis, then becomes medial, and ultimately becomes trans. Here, each stage possesses different sets of enzymes as it moves away from its starting point.
Instead of unloading its protein content into another cisterna, it modifies the protein until it reaches its final “mature” state. Therefore, the order of movement of proteins through the Golgi apparatus is from cis to trans.
The Golgi body primarily functions as the packaging center of the cell. It sorts the proteins coming from the ER and then tags them to their destination sites.
Proteins and lipids undergo further modifications in the Golgi apparatus. Short chains of sugar molecules might get added or detached, or phosphate groups attached as tags.
Finally, the modified proteins are sorted depending on markers such as amino acid sequences and chemical tags and then packaged into vesicles that bud from the trans face of the Golgi. Some of these vesicles transport their contents to other cell parts like lysosomes or vacuoles for further usages. Others fuse with the membrane, delivering membrane-anchored proteins.
4) Lysosomes
A lysosome refers to the single membrane-bound cell organelle that contains digestive enzymes and acts as the organelle-recycling facility of an animal cell. Lysosomes are an integral part of the endomembrane system, as some vesicles that leave the Golgi are bound for them. They form by the fusion of budded-off vesicles from the trans-Golgi.
Lysosomes contain acid hydrolases which require an acidic environment for optimal performance. They maintain an acidic environment by maintaining a pH of 5.0 inside the organelle. These hydrolytic enzymes are used for digesting worn-out organelles, food particles, proteins, nucleic acids, lipids, complex sugars, misfolded proteins, and engulfed viruses or prokaryotes. The lysosomes are also involved in the repair of the cell membrane. If the cell is beyond repair, the lysosomes’ self-destruct’ or ‘autolysis’ gives them the name ‘suicidal bags.’
Lysosomes carry out intracellular digestion by phagocytosis. In this process, they fuse with a vacuole and release their enzymes into the vacuole. Through phagocytosis, sugars, amino acids, and various other cell nutrients pass into the cytosol.
Lysosomes also recycle the cell’s obsolete organelles by a process called autophagy using hydrolytic enzymes.
5) Endosomes
Like lysosomes, endosomes are also single membrane-bound cytoplasmic structures through which molecules that have been taken into the cell via endocytosis travel to the lysosome for ‘digestion.’
6) Transport Vesicles
Vesicles are sac-like small membrane-bound transport units that ship molecules between different cytoplasmic compartments. Most vesicles primarily transfer the membranes assembled in the ER to the Golgi apparatus and then from there to various other locations.
Every cell has several types of vesicles, each having a distinct protein configuration and origin. For instance, the ER vesicles transport vesicles that bud off from the ER to translocate the protein cargo to the cis face of the Golgi apparatus. Another example is the Golgi vesicle, which pinches off the Golgi to transport its content externally (via exocytosis or secretory vesicles) or internally (via intracellular transport).
The target membrane of the protein containing vesicle contains a marker on its cytosolic surface that compliments the proteins on the vesicle traveling to the membrane. Once the vesicle finds the particular membrane, they fuse.
There are three types of vesicles, each having different functions: clathrin-coated, COPI-coated, and COPII-coated vesicles. For instance, clathrin-coated vesicles transport substances between the Golgi apparatus and the plasma membrane, whereas COPI and COPII-coated vesicles frequently transport cargo between the ER and the Golgi apparatus.
7) Cell (Plasma) Membrane
The cell membrane (plasma membrane) is a protective phospholipid bilayer membrane that separates the cell from its surroundings and regulates the transport of molecules and signals into and out of the cell. It also has embedded proteins that perform its functions. The plasma membrane is not a rigid structure; it is quasifluid. So, the molecules that compose the membrane are capable of lateral movement. Small molecules such as carbon dioxide, water, and oxygen can pass through the membrane freely by diffusion or osmosis. In contrast, cells actively transport large molecules.
The cell membrane is involved in cell-cell contact and signaling. It also helps in the exchange of materials in and out of the cell by endocytosis and exocytosis.
The cell membrane has multiple functions: transporting nutrients into the cell, removing waste, preventing the entry of harmful materials, maintaining the pH of the cytoplasm, and preserving the osmotic pressure of the cell interior. These functions are done by transport proteins that are very selective in their action and use ATP hydrolysis to pump or actively exchange materials against their concentration gradients.
The glycoproteins on the membrane assist the cell in recognizing other cells to exchange metabolites and form tissues. On the other hand, proteins on the plasma membrane allow attachment to the cytoskeleton and extracellular matrix, which maintains cell shape and fixes the location of membrane proteins. The plasma membrane also contains necessary enzymes that catalyze reactions. Also, receptor proteins on the membrane have a complementary shape to a chemical messenger, resulting in various cellular responses.
FAQs
Ans. Yes, the nucleus is a part of the endomembrane system.
Ans. Lysosomes are considered part of the endomembrane system because they contain many hydrolytic enzymes such as lipases, proteases, carbohydrates, and nucleases capable of digesting carbohydrates, proteins, fats, nucleic acids.
Ans. Peroxisomes are not considered part of the endomembrane system because they do not have any coordination or transaction of vesicles with the other components of the system.
Ans. Yes, the nuclear membrane is a part of the endomembrane system.
-
References
Article was last reviewed on Friday, February 17, 2023