Tonicity
Cells must remain turgid to function correctly. A well-watered plant grows and reproduces, producing fruits and flowers. In contrast, a plant not watered for a few days wilts and die. It happens because water moves out of the cell, causing the cell to lose turgidity.
Water moves in or out of the cell based on the solution’s solute concentration in which the cell remains suspended. Osmosis is the process that defines water’s net movement across a semipermeable membrane from a low solute concentration region to a high concentration region.
What is Tonicity
Tonicity is the extracellular solution’s ability to make water move inside or outside the cell by the process of osmosis. It measures the amount of solute dissolved in a specific amount of the solution, also known as the solution’s osmolarity.
Tonicity and Osmolarity
A low osmolarity solution has a greater number of water molecules compared to the number of solute particles. In contrast, a solution with high osmolarity has fewer water molecules than the solute particles. Tonicity also takes into account the permeability of the cell membrane to those solutes.
When solutions of two different osmolarities are separated by a selectively permeable membrane, which is permeable to water but not to solutes, water moves from the side of the membrane with low osmolarity to the side with high osmolarity. In other words, osmolarity lays down the direction of water flow in the cell membrane.
Types of Tonicity in Solution
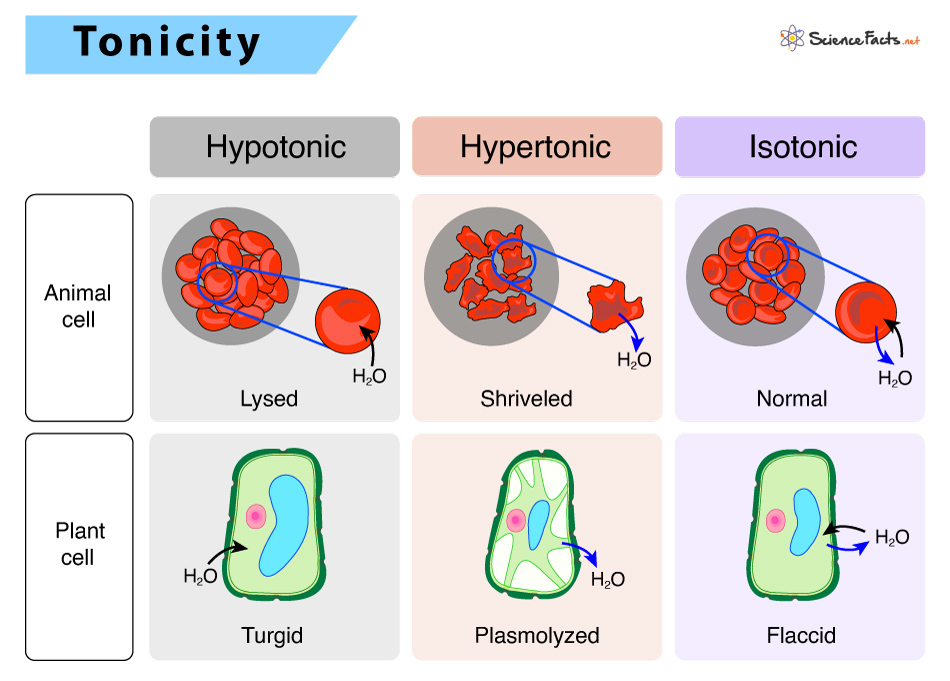
Based on the relative osmolarity of a cell to the osmolarity of the extracellular fluid, solutions can be hypertonic, hypotonic, and isotonic
1) Hypotonic Solutions
In a hypotonic environment, the extracellular fluid has lower osmolarity or solute concentration than the cell’s fluid.
In living systems, the cytoplasm is the point of reference. The prefix ‘hypo’ refers to the solution outside the cell, which has a low osmolarity than the cell cytoplasm. Thus, the extracellular fluid has a higher concentration of water than inside the cell.
If a cell is placed in a hypotonic solution (example: tap water), there will be a net flow of water inside the cell from the extracellular environment, causing the cell to increase in volume or expand.
2) Hypertonic Solutions
In a hypertonic situation, the prefix ‘hyper’ refers to the solution outside the cell, which has a high osmolarity compared to the cell cytoplasm. Thus, the extracellular fluid has a higher concentration of solute than inside the cell.
If a cell is placed in a hypertonic solution (example: seawater), there will be a net flow of water outside the cell, causing the cell to lose volume or shrink.
3) Isotonic Solutions
In an isotonic environment, the extracellular fluid has the same osmolarity as inside the cell.
When a cell is placed in an isotonic solution, there will be no net movement of water in or out of the cell, and the cell volume will remain the same. In other words, the amount of water that moves inside the cell is equal to the amount that moves out.
Examples of Tonicity in Living Systems
In Animals
For maintaining homeostasis, cells of our body, such as red blood cells, exist in an isotonic environment. If placed in a hypotonic solution, red blood cells will inflate, causing the cell to burst open. In contrast, when placed in a hypertonic environment, the cell will shrink, making the cytoplasm dense and highly concentrated. It may even cause cell death.
In Plants
Here, the hypotonic environment is ideal for a cell to function. For the cell (plasma) membrane to function correctly without being lysed, it expands to the required limit. The cytoplasm in plants is generally slightly hypertonic to the extracellular environment, allowing water to enter the cell until its internal turgor pressure prevents further influx.
When a plant is not watered, the extracellular fluid becomes hypertonic, causing water to leave the plant cell. It results in a loss of turgor pressure, causing wilting. In a hypertonic environment, the cell membrane detaches from the cell wall, causing the cell to constrict, a state known as plasmolysis.
Organisms such as paramecium and amoebae, lacking a cell wall, possess specialized structures called contractile vacuoles that maintain osmotic pressure within the cell. It collects excess water from the cell and pumps it out, protecting the cell from lysis.
FAQs
Ans. Osmolarity is the measure of solute concentration per unit volume of the solvent. In contrast, tonicity measures the osmotic pressure gradient between two solutions separated by a selectively permeable membrane. Thus, tonicity is only influenced by solutes that are prevented by the membrane to pass through.
-
References
Article was last reviewed on Thursday, February 2, 2023