Why Does Ice Float on Water
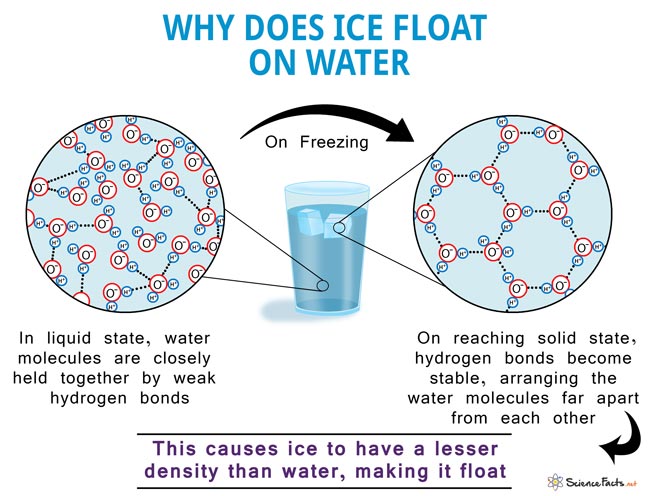
It is common for us to observe ice cubes floating when placed in a glass of water, and icebergs floating on the surface seas and oceans. However, this is a peculiar behavior as solid matter usually sinks in liquid. But ice, or water in its solid state, floats in liquid water for having a lesser density.
What Makes Ice Less Dense Than Liquid Water
Like all other substances, when cooled, water reduces in volume causing the molecules to get closer to each other, increasing the density. But as it gets colder than 4oC, water starts to expand causing a decrease in density. On further cooling, it turns into ice at 0oC, becoming 9% less dense than liquid water.
Understanding the Reason Behind This
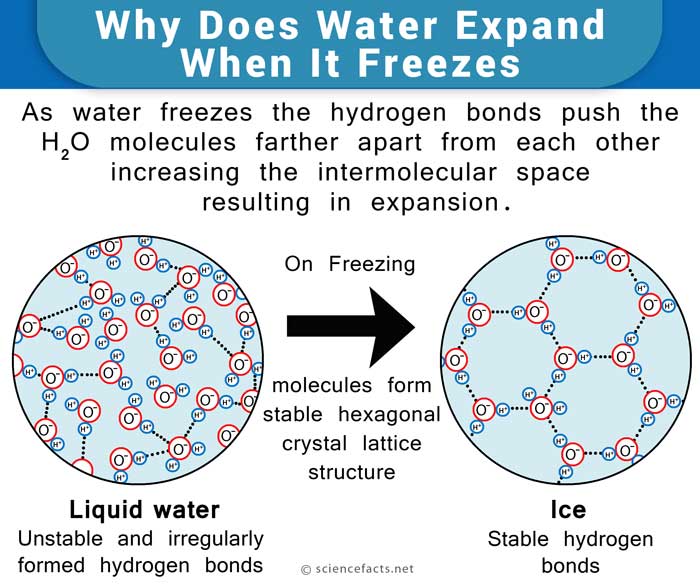
Water is made up of 2 atoms of hydrogen and one of oxygen. The slightly negatively charged oxygen atom of a water molecule is attracted towards the positively charged hydrogen atoms of neighboring water molecules, giving rise to weak hydrogen bonds between them.
In the liquid state, all the water molecules are loosely held together and are free to move. On cooling, the molecules try to come closer, increasing the density. But once the temperature drops below 4oC, the repelling nature of negative oxygen atoms of the neighboring molecules prevent them from coming any closer. That is when its density starts to fall. As it reaches the solid state (ice), the hydrogen bonds become stable and arrange themselves in such a way that the adjacent oxygen atoms are spread far apart from each other, leaving a lot of space between the molecules. This causes ice to have a higher volume and lesser density than water, making it float.
FAQs
1. Why does ice sink in alcohol?
– Alcohol has a lower density than ice, which makes it sink.
2. Does heavy water ice float or sink in ordinary water?
– Heavy water ice sinks in regular water as it has a higher density (but it will float in heavy water).
Article was last reviewed on Tuesday, December 10, 2019