Visible Light
Definition: What is Visible Light?
Light is a form of electromagnetic wave and constitutes a small part of the electromagnetic spectrum that is visible to the human eye. Hence, it is called visible light. Since visible light is a form of a wave, it is defined by its characteristic wavelength and frequency. The visible light spectrum is located within a wavelength range of 380 to 740 nanometers (nm) or a frequency range of 405 to 790 terahertz (THz).
Facts and Characteristics of Visible Light
- Consists of seven colors – violet, indigo, blue, green, yellow, orange, and red. Collectively known as VIBGYOR.
- Speed is constant, i.e., 3 X 108 m/s in the air
- Speed reduces to 2.25 X 108 m/s in water and 2 X 108 m/s in glass
- It can be measured by the unit of the lumen, which is the total quantity of visible light emitted by a source per unit of time. Another unit is candela, which gives the wavelength-weighted power or luminous intensity.
- Visible light usually is not dangerous or harmful. However, if the light is intense, then it can damage the receptor cells in the eye, causing temporary or permanent blindness.
Examples of Visible Light
- Sunlight
- The light emitted by some lasers
- Incandescent light bulbs, torchlight, flashlight, headlamps, fluorescent and neon lights
- The red, amber, and green traffic lights
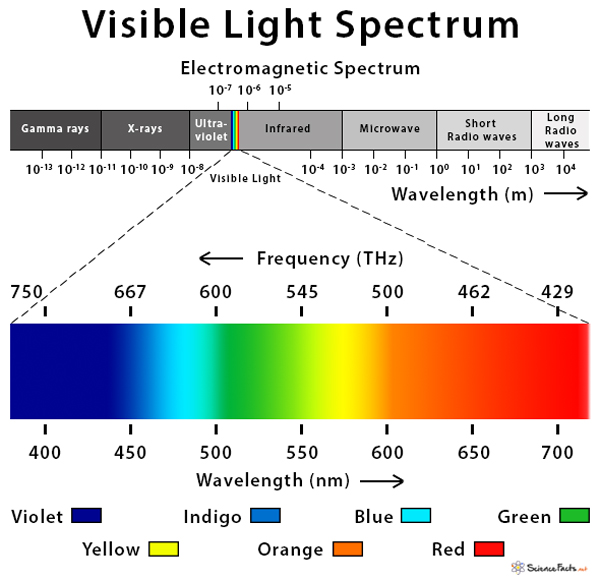
Visible Light Spectrum
Visible light falls in between the ultraviolet and the infrared regions of the electromagnetic spectrum.
Wavelength and Frequency of Visible Light
Each color of the visible light spectrum has its distinct wavelength and frequency.
| Color | Wavelength (nm) | Frequency (THz) |
| Violet | 380 – 450 | 680 – 790 |
| Indigo | 450 – 485 | 620 – 680 |
| Blue | 485 – 500 | 600 – 620 |
| Green | 500 – 565 | 530 – 600 |
| Yellow | 565 – 590 | 510 – 530 |
| Orange | 590 – 625 | 480 – 510 |
| Red | 625 – 740 | 405 – 480 |
Uses and Applications of Visible Light
Visible light can be used in many ways. The main benefit of visible light is that it allows us to see objects, thereby leading to many applications. Some of them are mentioned below:
- For performing scientific experiments by scientists, researchers, technicians, and students
- Lasers emitting visible light are used in laser pointers, laser surgery, and cutting of metals
- Light-emitting diodes (LED) display visible lights and are used in many electronic devices
- Astronomers use visible light emitted by distant objects like planets and moons to study their properties and features
- Also, sunlight, the primary source of visible light on the earth, has many benefits. These include a) Source of vitamin D, b) Kills bacteria, c) Builds the immune system, and d) Photosynthesis by plants.
FAQs
Ans. Visible light occupies a tiny portion of the electromagnetic spectrum – about 0.0035 percent.
Ans. The shortest wavelength with the visible spectrum is 380 nm. Violet has the shortest wavelength.
Ans. The longest wavelength with the visible spectrum is 740 nm. Red has the longest wavelength.
Ans. Violet has the highest frequency.
Ans. The sun produces white light, which is visible to the human eye. White light is a combination of all seven colors of the visible light spectrum.
Ans. The energy of visible light is electromagnetic.
Ans. The energy of the visible light spectrum ranges from 1.65 to 3.10 eV. The violet color has the highest energy.
Ans. It is not possible to see molecules with visible light, because the wavelengths of visible light are very much longer than the usual molecular dimensions.
Article was last reviewed on Wednesday, March 9, 2022








Good info
How do you block these dang led headlights. Do you need to wear a welders mask while driving night and day?
You could consider using polarized sunglasses for driving, which help cut down on the intensity of light