Valence Electrons
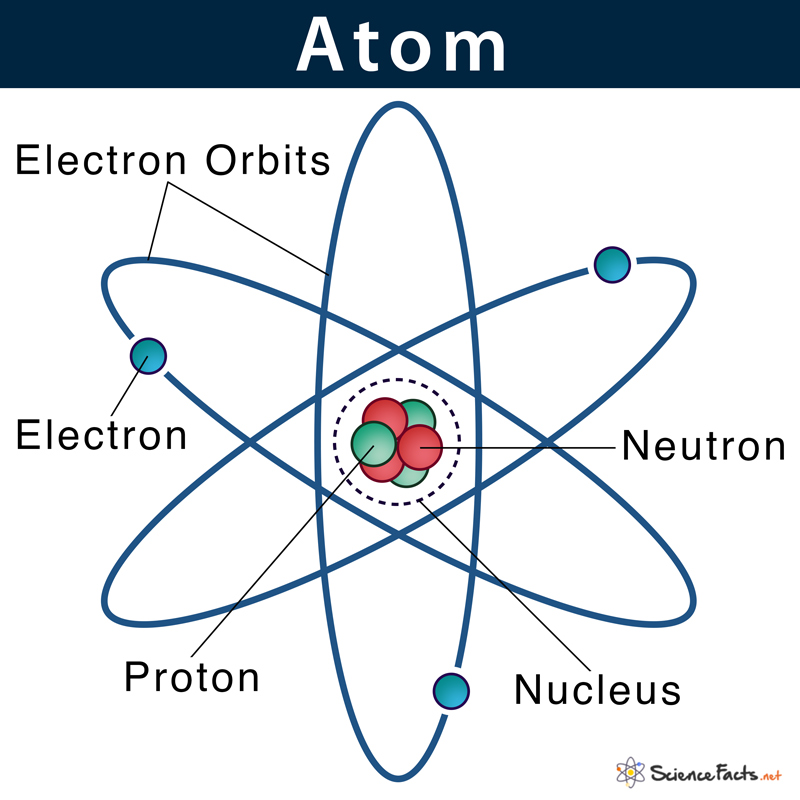
Valence electrons are located in an atomic nucleus’s outermost shell (the valence shell). As they are farthest from the nucleus, they are loosely attached to it. They actively participate in all its reactions by forming bonds with the valence electron of the adjacent atoms.
Why are Valence Electrons Important
The number of valence electrons determines the reactivity of the atom. They also determine the atom’s electronegativity, electron affinity, and ionization energy, which lead to things such as covalent and ionic bonds.
Atoms are most stable if they possess a filled valence shell, with each shell having 8 valence electrons. An exception to this phenomenon is hydrogen and helium, which become stable with 2 electrons in their outermost shell.
Characteristics of Valence Electrons
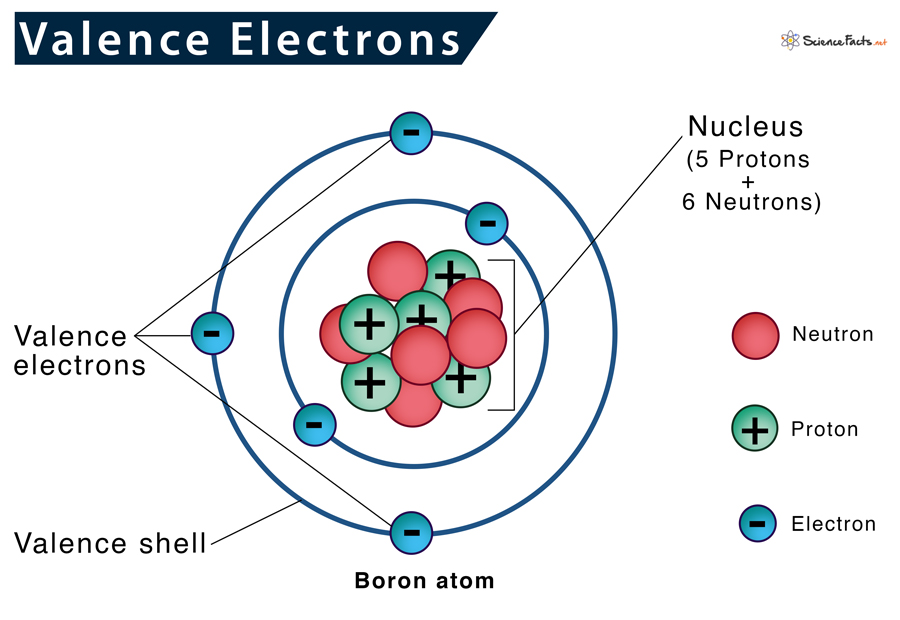
- They exist only in the outermost electron shell. However, they can exist in the inner shell of a transition metal.
- Atoms with valence shells filled with electrons (octet or duplet) are chemically inert.
- They absorb or release energy as small packets of energy (photons).
- They categorize elements into metal, non-metal, or metalloid based on the electrical conductivity of an element.
How to Find Valence Electrons in an Element
There are two ways of calculating the number of valence electrons in an element.
1. By Using Periodic Table
It is the most widely used method to determine the number of valence electrons in an element. Here, we just refer to the periodic table and search for the position of the element in it. As we proceed downwards in a group, the numbers of valence electrons are same, although the number of shells increases. In contrast, the number of valence electrons across a period increases by one as we move left to right of a period.
Exception: As discussed, the period number indicates the number of shells, whereas the group number specifies the valence electron number in the outermost shell of an atom. However, this only holds for the main group elements, groups 1-2 and 13-18. The rule applies to the transition and inner transition elements in groups 3-12.
Valence Electrons Periodic Table Chart
The table below depicts the number of valence electrons in the different groups of the periodic table:
| Periodic Table Group | Valence Electrons |
|---|---|
| Group 1 (I) – Alkali metals | 1 |
| Group 2 (II) – Alkaline earth metals | 2 |
| Group 13 (III) – Boron group | 3 |
| Group 14 (IV) – Carbon group | 4 |
| Group 15 (V) – Nitrogen group | 5 |
| Group 16 (VI) – Oxygen group | 6 |
| Group 17 (VII) – Halogens | 7 |
| Group 18 (VIII or 0) – Noble gases | 8 |
2. By Using Electronic Configuration of the Element
Within the shells, electrons occupy a special place called atomic orbitals. The arrangement of electrons in such orbitals is known as electronic configuration. The configuration of electrons gives a quick overview of the number of electrons present in the last shell. So, just by writing the electronic configuration of that element, we can quickly determine its number of valence electrons.
Here is a table representing the number of valence electrons of elements belonging to the second period and their electronic configuration.
| Name of the element | Symbol | Atomic Number | Electron Configuration | No. of valence electrons |
|---|---|---|---|---|
| Lithium | Li | 3 | 1s22s1 | 1 |
| Beryllium | Be | 4 | 1s22s2 | 2 |
| Boron | B | 5 | 1s22s22p1 | 3 |
| Carbon | C | 6 | 1s22s22p | 4 |
| Nitrogen | N | 7 | 1s22s22p3 | 5 |
| Oxygen | O | 8 | 1s22s22p4 | 6 |
| Fluorine | F | 9 | 1s22s22p5 | 7 |
| Neon | Ne | 10 | 1s22s22p6 | 8 |
Representing Valence Electrons
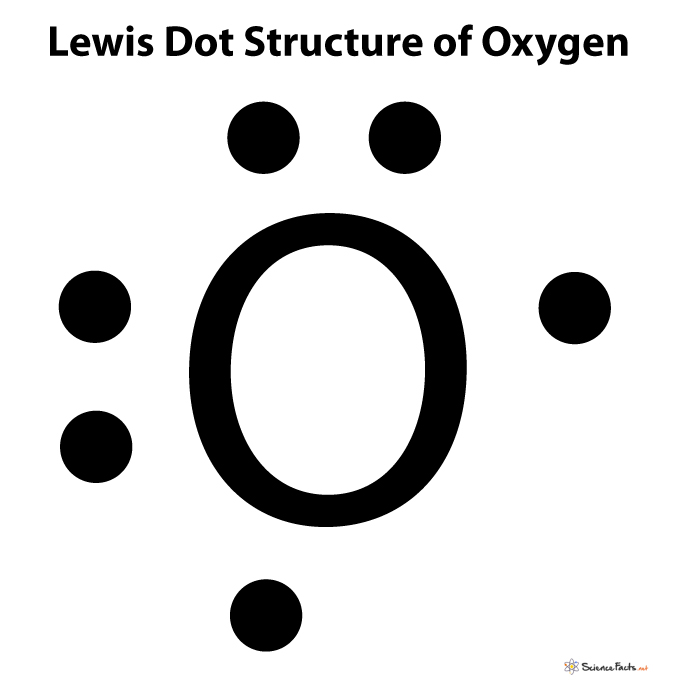
Lewis structure is an easier representation of the valence shell electrons in a molecule. Here, the atom is represented using a special Lewis symbol.
First, write the element’s chemical symbol, and then show its valence electrons surrounding it using dots. Show only the electrons residing in the valence shell as it solely determines the chemical reactivity of an atom.
For example, the Lewis symbol of oxygen depicts a ‘O’ surrounded by 6 valence electrons because oxygen has an electron configuration of 1s22s22p4.
-
References
Article was last reviewed on Thursday, February 2, 2023





I enjoy it very much